Which COVID-19 vaccine should I choose for my young child?
Let's break down some notable differences between Moderna and Pfizer
The CDC and FDA have officially authorized 2 COVID-19 vaccines for children over 6 months of age. On Saturday (6/18/22), CDC Director Dr. Rochelle Walensky made the votes of the CDC’s ACIP official, meaning that children can now receive either the Pfizer or the Moderna COVID-19 vaccines.
This means that COVID-19 vaccines will now be available for our youngest children (except kids younger than 6 mos). Many pediatrician offices will be setting up vaccine clinic days to roll these out. So of course, the obvious question is: which vaccine should I get for my child, Pfizer or Moderna?
The obvious answer if you only have access to one is whichever is available to you. However, some parents will have access to both, so that makes the decision a little trickier.
Both were unanimously supported by the FDA’s VRBPAC and the CDC’s ACIP – external committees tasked with reviewing clinical trial data.
Both are safe and effective, and led to the production of high levels of neutralizing antibodies similar to the control immunobridging groups (16-25 year olds).
Both protect against symptomatic and severe illness due to COVID-19.
There are some differences. While both use mRNA encoding the spike protein of SARS-CoV-2, formulations are not identical, so we cannot compare directly. But we can make generalizations.
Let’s discuss vaccine characteristics first.
Moderna is a 2-dose regimen, with each dose 25 micrograms of active ingredient (mRNA encoding spike protein). This means that it is fewer injections for your child, and shorter time to ‘full protection’ which broadly speaking, means 2 weeks after the final vaccination of a given series (this 2-week period is the average duration we observe full maturation of memory immunity).
Pfizer is a 3-dose regimen, with each dose 3 micrograms of active ingredient. This means more injections for your child, with a longer period to ‘full protection’. However, it also means smaller dosages per injection.
We also included the already authorized Pfizer vaccine for 5-11 year olds for comparison, as some parents have children soon to be turning 5. This has the shortest period to full protection of these 3, and is an intermediate dosage.
Remember though: while Moderna and Pfizer both use mRNA as an active ingredient, they are not identical in formulation or structure, so we cannot compare them identically - we can only make generalizations.
With regard to efficacy, this is a bit trickier to parse out. Pfizer reported vaccine efficacy against symptomatic COVID-19 of only about 28% after 2 doses, which was the motivation behind including a third dose in the primary vaccination series.
Remember, vaccine trials are balancing safety (tolerability) with immune response (immunogenicity). For pediatric trials, a de-escalation process is used, which means in Phase 1, researchers test dosages starting from the adult dosage and moving lower in dosage to find that optimcal balance. In this case, Pfizer used 1/10th of an adult dose in the young pediatric trials, compared to Moderna, which used 1/4th of their adult dose in young pediatric trials.
Okay, so Pfizer added a third dose in during their phase 3 trial, and report an efficacy against symptomatic COVID-19 of ~80%. However, we need to take this with a grain of salt. There were only a total of 10 COVID-19 cases after dose 3 was administered at the time the data were presented/reviewed (7 in placebo and 3 in vaccine arm). As such, this is a very small sample size, with a wide range of error. So we cannot definitively conclude this will be the real-world effectiveness of the Pfizer vaccine. More data will be collected as more COVID-19 cases occur in trial participants, and this efficacy will likely be adjusted (and will likely decline somewhat, just based on what we know about the vaccine effectiveness in other populations in the presence of Omicron).
Moderna reports an average efficacy against symptomatic COVID-19 of ~ 48%. Remember, data for both of these pediatric trials were collected during the Omicron period, and we know that Omicron is able to evade neutralizing antibodies better than the previous strains - however, vaccination still protects against severe illness, hospitalization, and death. This is in contrast to the 5-11 year trial data which was collected pre-Omicron, which is why reported efficacy amongst this group is apparently higher.
Ultimately, that’s why immunogenicity, or levels of neutralizing antibodies, are a useful metric here. Both Moderna and Pfizer led to production of high levels of neutralizing antibodies, and both were higher than the control groups used for normalization (which happened to be 18-25 year olds). As such, we can conclude that both elicit a robust immune response, even if we can’t definitively conclude the real-world effectiveness at this moment in time.
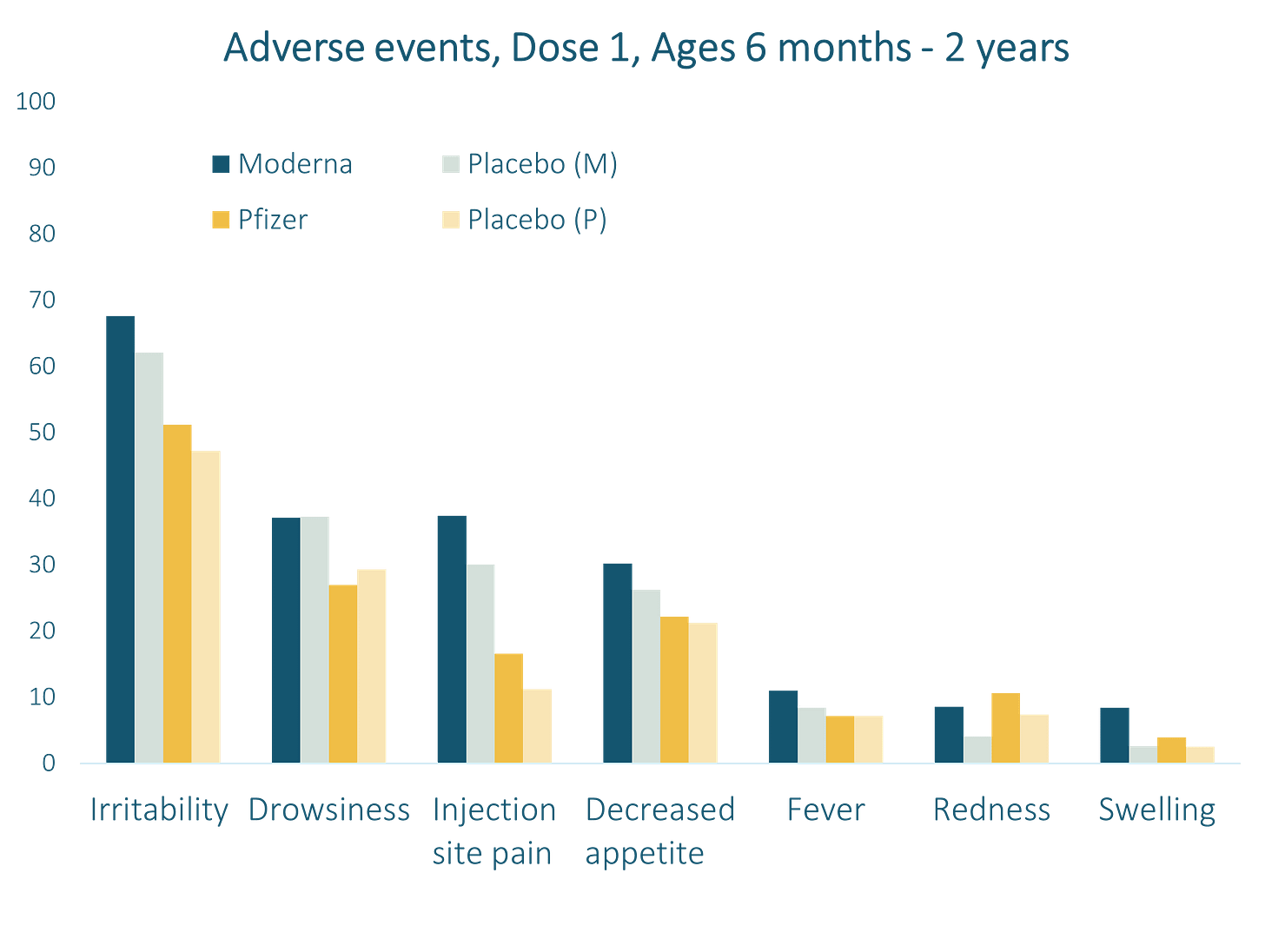
Let’s discuss adverse events, also called reactogenicity, next.
Broadly speaking, Moderna vaccination resulted in more adverse events among trial participants compared to Pfizer. This is likely due to the higher dosage of active ingredient among trial participants. However, these proportions of adverse events were similar (and in most cases, lower) than adverse events reported among 5-11 year olds in the Pfizer vaccine trial. Nearly all adverse events were mild and resolved within 1-2 days of vaccination across all groups.
When we compare the reported adverse events to placebo groups in both trials, it becomes apparent that there is a substantial nocebo effect, in both groups 6 months - 2 years, and 2-5 (or 6) years. Nocebo effects are a negative placebo effect, where someone who has actually received a placebo of a test treatment reports adverse events. These data are important to inform what are true adverse events, and what are potentially related to the physical act of injection, or have a potential psychological or psychosomatic link. As you can see, even amongst the most common reported adverse events, the number of those reported in placebo groups were very similar, and in some instances, exceeded the proportions reported in the vaccine groups.
We can conclude that these vaccines are extremely well-tolerated amongst this age group.
It is likely that Moderna will implement a third dose in the near future, particularly as new sub-variants of Omicron are predominant in the US, which require higher levels of neutralizing antibodies to offer optimal protection against. In addition, both companies are working on variant-specific boosters for all age groups, but these will likely not be available for at the very least, several months. Ultimately, you don’t want to wait to see if these will be available before offering your young children protection.
We must also remember that trial populations are not identical. Different children were enrolled in each, and behaviors, COVID-19 exposures, and demographics vary. As such, we cannot compare each data set directly, even though they both enrolled children 5 and younger.
Ultimately, there is no right or wrong decision. Consider these differences when making your choice, but remember, with Omicron subvariants circulating, the best choice is the vaccine you have access to the soonest.
Additional contributors:
Additional Resources:
https://www.fda.gov/advisory-committees/advisory-committee-calendar/vaccines-and-related-biological-products-advisory-committee-june-14-15-2022-meeting-announcement?fbclid=IwAR15pqFRr-FG3UsKXcO5SOZewBjbjrnhLqZ49bs53ViYm6RHW2lt_zMbecA https://www.cdc.gov/vaccines/hcp/acip-recs/vacc-specific/covid-19.html https://www.cdc.gov/vaccines/acip/meetings/slides-2022-06-17.html https://www.cdc.gov/vaccines/acip/meetings/slides-2022-06-18.html https://www.cdc.gov/media/releases/2022/s0618-children-vaccine.html







I have access to both vaccines. My child is 20 months old. I know both vaccines are good and effective but is there one that is preferable over the other if you have the choice?