Remdesivir receives FDA approval for COVID-19 treatment in young children
This marks the first approved treatment for children under 12
3 minute read
Yesterday, the FDA expanded approval of the COVID-19 treatment Veklury (remdesivir) to include pediatric patients 28 days of age and older weighing at least 3.5 kilograms (about 7 pounds) diagnosed with COVID-19, who are either: (1) hospitalized, or (2) not hospitalized with mild-to-moderate COVID-19 and are at high risk for progression to severe COVID-19, including hospitalization or death. This makes Veklury the first approved COVID-19 treatment for children under 12 years.
Prior to this, remdesivir was fully approved for certain adults and pediatric patients (12 years of age and older who weigh at least 40 kilograms, about 88 pounds), and was previously authorized under EUA for pediatric patients younger than 12 weighing at least 3.5 kg (as of January 21, 2022).
Remdesivir is considered a broad-spectrum antiviral, and it works by interfering with viral replication. It is a nucleoside analog - which means that it mimics the building blocks of viral genomes, called nucleotides.
When a virus replicates, it has to make genome copies (either RNA or DNA, depending on the type of virus). Instead of using the real nucleotide bases to make correct viral RNAs (in the case of SARS-CoV-2), the remdesivir molecules (the incorrect analog) is incorporated. This incorporation leads to errors during the viral RNA replication, and in the case of SARS-CoV-2, leads to premature termination. Ultimately, this means that the virus is unable to make additional correct copies of RNA to produce new virus particles, and as a result, inhibits viral replication. This can lead to reduction of illness severity, as viral load will be reduced, and this can be critical for limiting disease burden and mortality especially among high risk individuals.
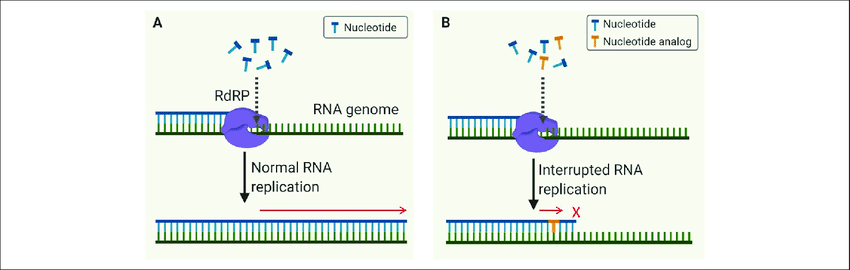
Remdesivir has also been investigated for other viral infections, including Hepatitis C virus, Ebola virus, and Marburg virus.
Just as a reminder: treatment is NEVER a substitute for vaccination; if it’s possible to prevent illness, that is always preferred over treating an illness, as there are far fewer risks.
Over 11 billion doses of COVID-19 vaccines have been safely administered globally at this point in the pandemic. Unverified reports submitted to VAERS do not constitute evidence of injury caused by vaccines; any serious reports to VAERS are investigated to determine causality (that is, whether it is causation, correlation, or a falsified claim). It is also important to remember that over 6 million people have lost their lives due to COVID-19, and vaccines prevent those deaths.
We know many parents are waiting for vaccines to become available for children under 5. While we do not have any updates on that, it is a bit of good news that there are some treatments available should young children become very sick.
A real-world study on remdesivir was recently conducted by researchers at Johns Hopkins University, including data from over 43,000 patients hospitalized with COVID-19.
Overall, 74% of remdesivir-receiving patients saw improvement within 28 days (median time of 7 days) versus 68.3% of control patients (median time of 9 days). Remdesivir patients receiving either low-flow oxygen or no oxygen had significantly greater clinical improvement than control patients. Remdesivir also significantly reduced mortality in patients on low-flow oxygen, even when accounting for the effects of anti-inflammatory medications, such as dexamethasone. Of patients on low-flow oxygen, the 28-day mortality rate of remdesivir recipients was 4.1% lower than that of the control patients.
Taken together, having remdesivir as a treatment option for high risk children is another important tool in our toolkit, especially as many of those children cannot be vaccinated yet. This approval will further contribute to reducing disease and mortality burden as we continue to combat the ongoing pandemic.
Additional contributors:


