More evidence demonstrating safety of COVID-19 boosters
Data from VAERS and V-safe indicate boosters are well-tolerated
5 minute read
A recent Morbidity and Mortality Weekly Report (MMWR) from the CDC provides more safety information following the administration of COVID-19 booster vaccines. Reports from both V-safe and VAERS were analyzed from September 22, 2021- February 6, 2022 in an ongoing effort to monitor the continued safety of COVID-19 vaccines. Remember, V-Safe and VAERS are both voluntary reporting systems that don’t require evidence of causality in order to file an adverse event report. During this period, 721,562 reported were filed among the 82.6 million booster doses administered during this period. This equates to less than 1% of people who received a booster reporting any adverse side effects. This provides further evidence of the incredibly safety of COVID-19 vaccines, specifically booster doses.
Boosters were given to people who received every type of the currently approved COVID-19 vaccines in the US (Pfizer, Moderna, and J&J).
The CDC analyzed reactions following booster doses with the help of two programs: V-Safe and VAERS. The first program, V-Safe, is a voluntary smartphone-based survey surveillance system and VAERS (Vaccine Adverse Event Reporting System) is a passive and voluntary vaccine safety surveillance system managed by the CDC and FDA. We’ve previously discussed limitations of VAERS. VAERS reports can be submitted by anyone, and require no evidence of causality, or even evidence that the person filing a report received a vaccine or was a healthcare provider of a vaccine recipient. Of all the reports filed, 88.8% received the same booster as their primary vaccination series.
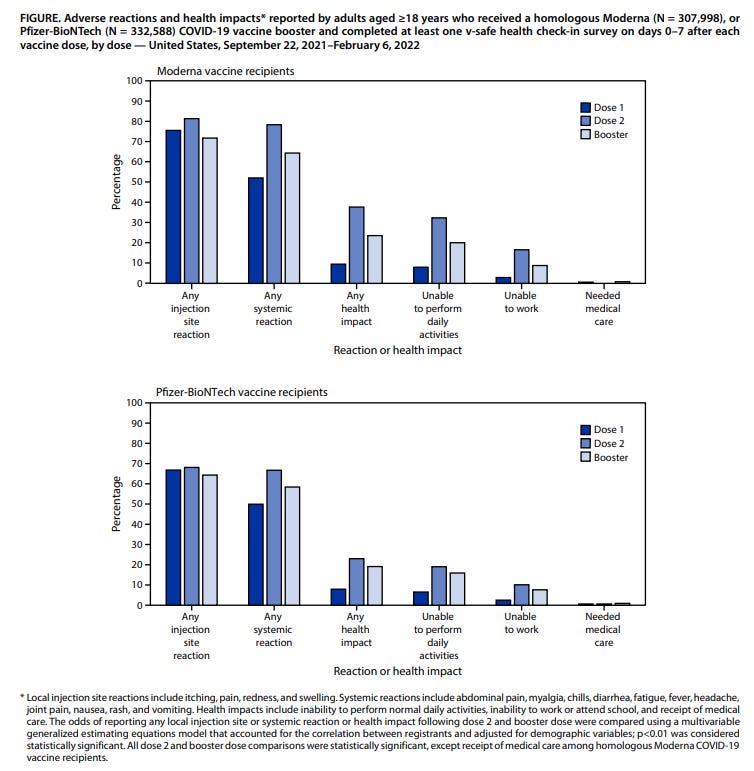
V-Safe is a unique data collection service that is able to track individuals over time and as they receive additional vaccine doses, since reports and data are linked to each individual’s phone number. As such, reports from a single person are able to be compared following receipt of different vaccine doses. The results of the V-Safe program demonstrated that of those who completed V-Safe check-ins following both their primary vaccination and booster doses, those who received the same booster as their initial primary vaccine series (called homologous prime-boost), appeared to have fewer reactions following their booster. Among 307,998 individuals who reported a homologous Moderna booster, reactions were less frequently reported during the week following booster (64.4%) than following dose 2 (78.4%). Among 332,588 registrants who reported a homologous Pfizer-BioNTech booster, reactions were also reported less frequently following the booster (58.4%) than following dose 2 (66.7%). These data are able to be monitored because V-Safe is linked to each individual through their phone number, and texts are sent periodically to check in with those who enroll. Remember, these numbers are based on the people who completed V-Safe check-ins, not the total number of vaccines administered. People are more likely to complete a check-in if they experienced a perceived adverse event.
The VAERS program received 39,286 reports of adverse events after the booster (92.4% are categorized as non-serious reactions; 7.6% serious events). VAERS reports are classified as serious if there are any reports of hospitalization, prolongation of hospitalization, life-threatening illness, permanent disability, congenital anomaly or birth defect, or death. Medical care was rarely sought for all of these reactions and myocarditis showed to be even more rare with only 37 cases reported to VAERS. There is one death report in VAERS attributed to myocarditis, but this is an ongoing investigation to determine causality (as is the case for all serious reports received by VAERS).
This study has four potential limitations.
First, V-Safe is a voluntary program, so it may not be representative of the entire vaccination program. It is dependent on people completing their V-Safe check-ins that are sent to their cell phone following each of their vaccine doses. Since people are more likely to complete something like that when they’ve had a perceived negative experience (as opposed to when they’ve experienced nothing), this may skew the data being collected.
Second, VAERS is a passive surveillance system and subject to reporting biases. Anything can be reported to VAERS regardless of whether it is truly linked to a vaccination, and there is no requirement or burden to prove causality. People have reported deaths on VAERS even if the death was not caused by the vaccine. VAERS accepts all reports and does not filter them; therefore, the numbers may not always be accurate.
Due to the low number of people receiving a J&J booster following an mRNA primary COVID-19 vaccine, the data were insufficient to assess this combination of heterologous prime-boost.
Finally, as any serious reports filed in VAERS are flagged for ongoing investigation, the reported instances of myocarditis are subject to change depending on the outcome of these investigations.
The CDC’s Advisory Committee on Immunization Practices (ACIP) recommends that all people 12 or older receive a COVID-19 booster dose at least 5 months following dose 2 of an mRNA vaccine.
Following this analysis, the data demonstrate that the booster vaccines are well-tolerated and have an incredibly high safety profile. Indeed, reactions following a COVID-19 booster are less common than following the second dose in the primary series. Less than 1% of those that received a booster dose during this period reported any adverse effects.
The CDC and FDA will continue to monitor vaccine safety and provide updates, as always, but this should give you confidence if you have not yet gotten your booster to do so. Currently, only 45.7% of eligible people have gotten their boosters, so it is important to do so if you haven’t!
Additional contributors: