COVID-19: Booster dose eligibility and state of affairs
Cases are rising, Omicron subvariants are predominant, and booster doses protect against illness and death
5 minute read
A few days ago we addressed a common question about whether, if eligible, you should get your COVID-19 booster, or if you should wait. And that of course, prompted some questions about which individuals are authorized for additional vaccine doses.
Let’s discuss what the current status is for boosters for different populations.
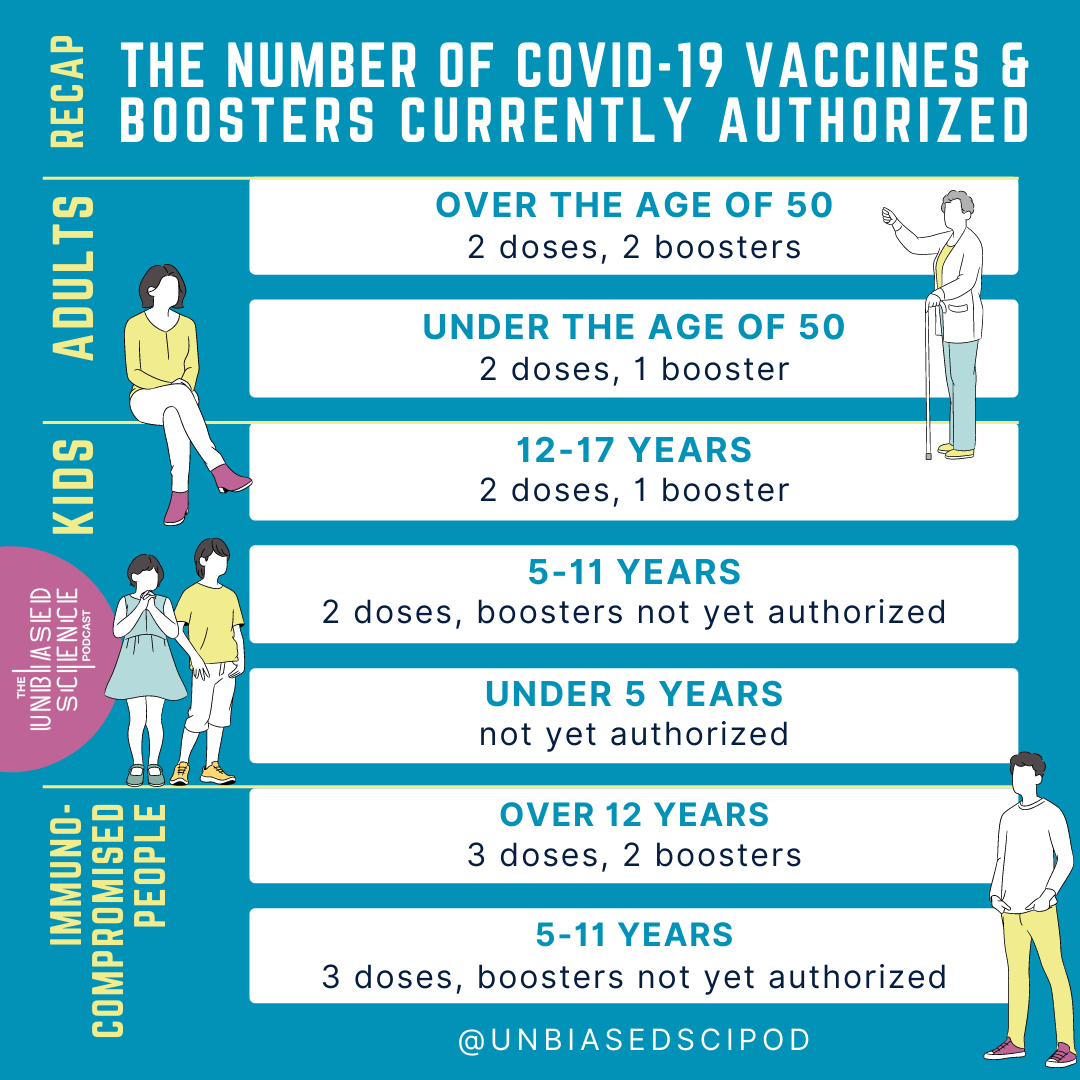
If you were an mRNA vaccine recipient (Moderna or Pfizer) and you are over 50 years, you are eligible for 2 doses (considered the primary series) and 2 boosters. The first booster of either Pfizer-BioNTech or Moderna COVID-19 vaccine is recommended at least 5 months after the final dose in the primary series
Adults ages 50 years and older can receive a second booster dose of either Pfizer-BioNTech or Moderna COVID-19 vaccine at least 4 months after the 1st booster.
For adults 18-50, you are eligible for the primary 2-dose vaccination series and a single booster administered 5 months after completion of your primary vaccination.
For kids 12-17, you are eligible for a Pfizer-BioNTech primary 2-dose vaccination series and a single booster 5 months after your primary vaccination.
For children 5-11, the primary vaccination series (2 doses, Pfizer-BioNTech) is authorized. No booster doses are currently authorized for this age group.
And yes, the case is still that children under 5 do not yet have any vaccines available to them, although Moderna has completed it’s submission of data to the FDA. We are expecting updates about eligibility for kids under 5 in June currently.
If you are immunocompromised and are over 12 years old, there is an additional vaccine dose added to the primary series. As such, you are eligible for 3 doses in a primary series. The second dose is administered either 3 weeks (Pfizer-BioNTech) or 4 weeks (Moderna) after the first dose, and the third dose is administered 28 days after the second dose. Following that, you’re eligible for a booster 3 months after your third dose. Recently, the FDA authorized a second booster dose for immunocompromised individuals at least 4 months after the initial booster dose.
For children 5-11 who are immunocompromised, the primary series also includes 3 doses; the second dose (Pfizer-BioNTech is the only authorized vaccine currently) administered at 21 days after the first dose, and the third dose 28 days after the second dose. There is no booster authorized for this group yet.
Who is considered immunocompromised?
People considered to be moderately or severely immunocompromised:
Those who have been receiving active cancer treatment for tumors or cancers of the blood
Those who have received a solid organ transplant and are taking medicine to suppress the immune system
Those who have received CAR T cell therapy, stem cell transplants, or other immune cell therapies within the last 2 years
Those with moderate or severe primary immunodeficiency (such as DiGeorge syndrome, Wiskott-Aldrich syndrome, asplenic individuals)
Those with advanced or untreated HIV infection
Those undergoing treatment with high-dose corticosteroids or other drugs that may suppress their immune response
If you have questions about your immunocompetency status, talk to your healthcare provider.
What about people who received the J&J (Janssen) vaccine, which was only a single dose in the primary vaccination?
It is recommended you receive a second vaccine (an mRNA one, either Pfizer-BioNTech or Moderna) 2 months after your J&J vaccine. If you received a J&J dose as your second vaccine (booster), you should receive a second booster of an mRNA vaccine 4 months after your first J&J booster. Individuals 50 and older are also eligible for an additional booster 4 months after your first mRNA booster.
Currently, the FDA and CDC are preferentially recommending the mRNA COVID-19 vaccines over the J&J vaccine, due to the rare risk of vaccine-induced thrombotic thrombocytopenia (VITT), a blood clotting disorder coupled with low platelets.
However, if the mRNA vaccines are not an option for you, due to either lack of access or severe allergies to mRNA vaccine ingredients, the J&J vaccine is still available.
Currently, Omicron (BA.1) and the newer subvariants are the predominant strains of SARS-CoV-2 around the country. BA.2 a more infectious subvariant of Omicron, is estimated to account for 56.4% of new cases in the US. BA.2.12.1, another subvariant of Omicron, is estimated to account for 42.6% of new cases. These subvariants of BA.1 have fitness advantages compared to the parental variant, which is why they have rapidly become the dominant variants in the US. All of these Omicron subvariants have additional mutations in the spike protein of the virus that makes them more able to evade our immune responses and the protection offered by vaccination. As such, it is extremely important that you get boosted if eligible.
Omicron and the Omicron subvariants are better able to overcome immunity due to infection (natural immunity), so you can't assume you won't get infected if you're relying on natural immunity. Vaccination provides more robust protection than natural immunity (we have many posts on this so please check our post database). Indeed, reinfection shortly after BA.1 with BA.2 is rare, but still possible. 89% of reinfection cases within 60 days of initial infection with Omicron (BA.1) were among unvaccinated people.
Case rates are rising again, and hospitalization rates are trending upward as well. Currently, deaths are not increasing, but we do know there is a lag in that metric following cases and hospitalizations. It is also likely that we are underreporting case numbers due to the prevalence of at-home testing (and some individuals not testing at all). Wastewater surveillance gives a better picture of community levels in this instance, and these data also indicate substantial increases in virus spreading across the nation.
While we have a decent proportion of the population vaccinated, there is improvement to be made. Currently, only 66.4% of the population is considered fully vaccinated (primary series), and 49.4% of those eligible for booster doses have not received one yet.
If you are not eligible for an additional booster (or a booster yet) understand that the high transmissibility of Omicron subvariants may mean that you have reduced protection against infection outright, but vaccines are protective against severe illness and death. Remember: antibodies are not the full story behind vaccine-induced protection against illness.
With all of the emerging data demonstrating that vaccine-induced protection is superior to natural immunity, please consider getting vaccinated and boosted if you’re eligible. If you’re holding out on getting a COVID-19 booster in the hopes that a variant-specific vaccine/booster will soon be available, DO NOT WAIT. Even if variant-specific vaccines become available later this year, it is very unlikely that getting boosted now will make you ineligible for a vaccine in the future.




My doc specifically said to get the Pfizer booster as it's a full-strength dose. Moderna booster is half-dose.